Abstract
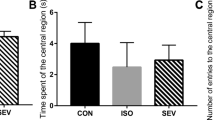
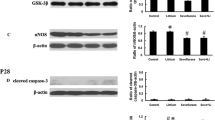
Long duration of anesthesia may induce toxicity in the developing brain. However, little is known about the effects of the combination of surgery and anesthesia on the developing brain. The mechanisms for the effects are not clear. To determine these effects, postnatal day 7 male and female Sprague-Dawley rats were exposed to 3% sevoflurane for 2 h with or without right common carotid exposure. Pyrrolidine dithiocarbamate (PDTC), an anti-inflammatory agent, was given 30 min before and 6 h after the carotid exposure. Anti-glial cell-derived neurotrophic factor (GDNF) antibody or GDNF was given at the end of sevoflurane exposure. We found that anesthesia-surgery induced learning and memory impairment assessed by Barnes maze and fear conditioning. Anesthesia-surgery also induced neuroinflammation and reduced the level of glial cell-derived neurotrophic factor (GDNF, 10.6 ± 0.6 pg/mg protein of control rats vs. 7.7 ± 0.4 pg/mg protein of anesthesia-surgery rats, n = 17, p = 0.007) and neurogenesis in the hippocampus. PDTC inhibited these surgical effects (GDNF level 9.7 ± 0.6 pg/mg protein of anesthesia-surgery plus PDTC rats, n = 17, p = 0.763 vs. control rats). Intracerebroventricular injection of an anti-GDNF antibody but not its heat-inactivated form induced learning and memory impairment in control rats. Intracerebroventricular injection of GDNF attenuated learning and memory impairment after anesthesia-surgery. We conclude that anesthesia-surgery in neonatal rats induces neuroinflammation, which then leads to a decreased level of GDNF and neurogenesis in the hippocampus and cognitive impairment. GDNF decrease plays an important role in anesthesia-surgery-induced cognitive impairment.
Key message
-
Anesthesia-surgery in neonatal rats induces neuroinflammation.
-
Neuroinflammation leads to decreased levels of GDNF.
-
Neuroinflammation reduces hippocampal neurogenesis and induces cognitive impairment.
-
GDNF decrease is important for anesthesia-surgery-induced cognitive impairment.







Similar content being viewed by others
References
Chiao S, Zuo Z (2014) A double-edged sword: volatile anesthetic effects on the neonatal brain. Brain Sci 4:273–294
Shen X, Dong Y, Xu Z, Wang H, Miao C, Soriano SG, Sun D, Baxter MG, Zhang Y, Xie Z (2013) Selective anesthesia-induced neuroinflammation in developing mouse brain and cognitive impairment. Anesthesiology 118:502–515
Lu Y, Huang Y, Jiang J, Hu R, Yang Y, Jiang H, Yan J (2016) Neuronal apoptosis may not contribute to the long-term cognitive dysfunction induced by a brief exposure to 2% sevoflurane in developing rats. Biomed Pharmacother 78:322–328
Yang B, Liang G, Khojasteh S, Wu Z, Yang W, Joseph D, Wei H (2014) Comparison of neurodegeneration and cognitive impairment in neonatal mice exposed to propofol or isoflurane. PLoS One 9:e99171
Cao L, Li L, Lin D, Zuo Z (2012) Isoflurane induces learning impairment that is mediated by interleukin 1beta in rodents. PLoS One 7:e51431
Cibelli M, Fidalgo AR, Terrando N, Ma D, Monaco C, Feldmann M, Takata M, Lever IJ, Nanchahal J, Fanselow MS et al (2010) Role of interleukin-1beta in postoperative cognitive dysfunction. Ann Neurol 68:360–368
Zhang J, Jiang W, Zuo Z (2014) Pyrrolidine dithiocarbamate attenuates surgery-induced neuroinflammation and cognitive dysfunction possibly via inhibition of nuclear factor kappaB. Neurosci 261:1–10
Fang F, Xue Z, Cang J (2012) Sevoflurane exposure in 7-day-old rats affects neurogenesis, neurodegeneration and neurocognitive function. Neurosci Bull 28:499–508
Fan D, Li J, Zheng B, Hua L, Zuo Z (2016) Enriched environment attenuates surgery-induced impairment of learning, memory, and neurogenesis possibly by preserving BDNF expression. Mol Neurbiol 53:344–354
van Praag H, Schinder AF, Christie BR, Toni N, Palmer TD, Gage FH (2002) Functional neurogenesis in the adult hippocampus. Nature 415:1030–1034
Shors TJ, Townsend DA, Zhao M, Kozorovitskiy Y, Gould E (2002) Neurogenesis may relate to some but not all types of hippocampal-dependent learning. Hippocampus 12:578–584
Sakamoto M, Imayoshi I, Ohtsuka T, Yamaguchi M, Mori K, Kageyama R (2011) Continuous neurogenesis in the adult forebrain is required for innate olfactory responses. Proc Natl Acad Sci U S A 108:8479–8484
Wang B, Jin K (2015) Current perspectives on the link between neuroinflammation and neurogenesis. Metab Brain Dis 30:355–365
Song C, Zhang Y, Dong Y (2013) Acute and subacute IL-1beta administrations differentially modulate neuroimmune and neurotrophic systems: possible implications for neuroprotection and neurodegeneration. J Neuroinflammation 10:59
Zhang J, Tan H, Jiang W, Zuo Z (2015) The choice of general anesthetics may not affect neuroinflammation and impairment of learning and memory after surgery in elderly rats. Journal Neuroimmune Pharm 10:179–189
Lee JJ, Li L, Jung H-H, Zuo Z (2008) Postconditioning with isoflurane reduced ischemia-induced brain injury in rats. Anesthesiology 108:1055–1062
Zhao P, Peng L, Li L, Xu X, Zuo Z (2007) Isoflurane preconditioning improves long-term neurologic outcome after hypoxic-ischemic brain injury in neonatal rats. Anesthesiology 107:963–970
Zhang J, Tan H, Jiang W, Zuo Z (2014) Amantadine alleviates postoperative cognitive dysfunction possibly by increasing glial cell line-derived neurotrophic factor in rats. Anesthesiology 121:773–785
Ellis P, Fagan BM, Magness ST, Hutton S, Taranova O, Hayashi S, McMahon A, Rao M, Pevny L (2004) SOX2, a persistent marker for multipotential neural stem cells derived from embryonic stem cells, the embryo or the adult. Dev Neurosci 26:148–165
Wolf HK, Buslei R, Schmidt-Kastner R, Schmidt-Kastner PK, Pietsch T, Wiestler OD, Blumcke I (1996) NeuN: a useful neuronal marker for diagnostic histopathology. J Histochem Cytochem 44:1167–1171
Zhang Z, Miyoshi Y, Lapchak PA, Collins F, Hilt D, Lebel C, Kryscio R, Gash DM (1997) Dose response to intraventricular glial cell line-derived neurotrophic factor administration in parkinsonian monkeys. J Pharmacol Exp Ther 282:1396–1401
Lapchak PA, Jiao S, Collins F, Miller PJ (1997) Glial cell line-derived neurotrophic factor: distribution and pharmacology in the rat following a bolus intraventricular injection. Brain Res 747:92–102
Zhou L, Wang Z, Zhou H, Liu T, Lu F, Wang S, Li J, Peng S, Zuo Z (2015) Neonatal exposure to sevoflurane may not cause learning and memory deficits and behavioral abnormality in the childhood of cynomolgus monkeys. Sci Rep 5:11145
Jiang Y, Tong D, Hofacer RD, Loepke AW, Lian Q, Danzer SC (2016) Long-term fate mapping to assess the impact of postnatal isoflurane exposure on hippocampal progenitor cell productivity. Anesthesiology 125:1159–1170
Wang Z, Zhao H, Peng S, Zuo Z (2013) Intranasal pyrrolidine dithiocarbamate decreases brain inflammatory mediators and provides neuroprotection after brain hypoxia-ischemia in neonatal rats. Exp Neurol 249:74–82
Li H, Yin J, Li L, Deng J, Feng C, Zuo Z (2013) Isoflurane postconditioning reduces ischemia-induced nuclear factor-kappaB activation and interleukin 1beta production to provide neuroprotection in rats and mice. Neurobiol Dis 54:216–224
Kobayashi T, Ahlenius H, Thored P, Kobayashi R, Kokaia Z, Lindvall O (2006) Intracerebral infusion of glial cell line-derived neurotrophic factor promotes striatal neurogenesis after stroke in adult rats. Stroke 37:2361–2367
Rocha SM, Cristovao AC, Campos FL, Fonseca CP, Baltazar G (2012) Astrocyte-derived GDNF is a potent inhibitor of microglial activation. Neurobiol Dis 47:407–415
Chen SH, Oyarzabal EA, Sung YF, Chu CH, Wang Q, Chen SL, Lu RB, Hong JS (2015) Microglial regulation of immunological and neuroprotective functions of astroglia. Glia 63:118–131
Stary CM, Sun X, Giffard RG (2015) Astrocytes protect against isoflurane neurotoxicity by buffering pro-brain-derived neurotrophic factor. Anesthesiology 123:810–819
Zuo Z (2001) Isoflurane enhances glutamate uptake via glutamate transporters in rat glial cells. Neuroreport 12:1077–1080
Robinson MB (1999) The family of sodium-dependent glutamate transporters: a focus on the GLT-1/EAAT2 subtypes. Neurochem Int 33:479–491
Yin J, Li H, Feng C, Zuo Z (2014) Inhibition of brain ischemia-caused notch activation in microglia may contribute to isoflurane postconditioning-induced neuroprotection. CNS NEUROL DISORD-DR 13:718–732
Yufune S, Satoh Y, Akai R, Yoshinaga Y, Kobayashi Y, Endo S, Kazama T (2016) Suppression of ERK phosphorylation through oxidative stress is involved in the mechanism underlying sevoflurane-induced toxicity in the developing brain. Sci Rep 6:21859
Tao G, Zhang J, Zhang L, Dong Y, Yu B, Crosby G, Culley DJ, Zhang Y, Xie Z (2014) Sevoflurane induces tau phosphorylation and glycogen synthase kinase 3beta activation in young mice. Anesthesiology 121:510–527
Snyder JS, Choe JS, Clifford MA, Jeurling SI, Hurley P, Brown A, Kamhi JF, Cameron HA (2009) Adult-born hippocampal neurons are more numerous, faster maturing, and more involved in behavior in rats than in mice. J Neurosci 29:14484–14495
Willing J, Drzewiecki CM, Cuenod BA, Cortes LR, Juraska JM (2016) A role for puberty in water maze performance in male and female rats. Behav Neurosci 130:422–427
Drzewiecki CM, Willing J, Juraska JM (2016) Synaptic number changes in the medial prefrontal cortex across adolescence in male and female rats: a role for pubertal onset. Synapse 70:361–368
Wilder RT, Flick RP, Sprung J, Katusic SK, Barbaresi WJ, Mickelson C, Gleich SJ, Schroeder DR, Weaver AL, Warner DO (2009) Early exposure to anesthesia and learning disabilities in a population-based birth cohort. Anesthesiology 110:796–804
DiMaggio C, Sun LS, Kakavouli A, Byrne MW, Li G (2009) A retrospective cohort study of the association of anesthesia and hernia repair surgery with behavioral and developmental disorders in young children. J Neurosurg Anesthesiol 21:286–291
Dimaggio C, Sun LS, Li G (2011) Early childhood exposure to anesthesia and risk of developmental and behavioral disorders in a sibling birth cohort. Anesth Analg 113:1143–1151
Flick RP, Katusic SK, Colligan RC, Wilder RT, Voigt RG, Olson MD, Sprung J, Weaver AL, Schroeder DR, Warner DO (2011) Cognitive and behavioral outcomes after early exposure to anesthesia and surgery. Pediatrics 128:e1053–e1061
Kalkman CJ, Peelen L, Moons KG, Veenhuizen M, Bruens M, Sinnema G, de Jong TP (2009) Behavior and development in children and age at the time of first anesthetic exposure. Anesthesiology 110:805–812
Bartels M, Althoff RR, Boomsma DI (2009) Anesthesia and cognitive performance in children: no evidence for a causal relationship. Twin Res Hum Genet 12:246–253
Hansen TG, Pedersen JK, Henneberg SW, Morton NS, Christensen K (2013) Educational outcome in adolescence following pyloric stenosis repair before 3 months of age: a nationwide cohort study. Paediatr Anaesth 23:883–890
Hansen TG, Pedersen JK, Henneberg SW, Pedersen DA, Murray JC, Morton NS, Christensen K (2011) Academic performance in adolescence after inguinal hernia repair in infancy: a nationwide cohort study. Anesthesiology 114:1076–1085
Davidson AJ, Disma N, de Graaff JC, Withington DE, Dorris L, Bell G, Stargatt R, Bellinger DC, Schuster T, Arnup SJ et al (2016) Neurodevelopmental outcome at 2 years of age after general anaesthesia and awake-regional anaesthesia in infancy (GAS): an international multicentre, randomised controlled trial. Lancet 387:239–250
Sun LS, Li G, Miller TL, Salorio C, Byrne MW, Bellinger DC, Ing C, Park R, Radcliffe J, Hays SR et al (2016) Association between a single general anesthesia exposure before age 36 months and neurocognitive outcomes in later childhood. JAMA 315:2312–2320
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Grant support
This study was supported by grants (GM098308 and NS094030 to Z Zuo) from the National Institutes of Health, Bethesda, MD, the Robert M. Epstein Professorship endowment, University of Virginia, Charlottesville, VA, and a grant (81641160) from National Natural Science Foundation of China, Beijing, China.
Conflict of interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Gui, L., Lei, X. & Zuo, Z. Decrease of glial cell-derived neurotrophic factor contributes to anesthesia- and surgery-induced learning and memory dysfunction in neonatal rats. J Mol Med 95, 369–379 (2017). https://doi.org/10.1007/s00109-017-1521-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-017-1521-9