Abstract
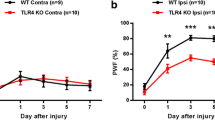
Microglia, rapidly activated following peripheral nerve injury (PNI), accumulate within the spinal cord and adopt inflammation that contributes to development and maintenance of neuropathic pain. Microglia express functional Toll-like receptors (TLRs), which play pivotal roles in regulating inflammatory processes. However, little is known about the role of TLR3 in regulating neuropathic pain after PNI. Here TLR3 expression and autophagy activation was assayed in dorsal root ganglions and in microglia following PNI by using realtime PCR, western blot and immunohistochemistry. The role of TLR3/autophagy signaling in regulating tactile allodynia was evaluated by assaying paw mechanical withdrawal threshold and cold allodynia after intrathecal administration of Poly (I:C) and 3-methyladenine (3-MA). We found that L5 spinal nerve ligation (SNL) induces the expression of TLR3 in dorsal root ganglions and in primary rat microglia at the mRNA and protein level. Meanwhile, L5 SNL results in an increased activation of autophagy, which contributes to microglial activation and subsequent inflammatory response. Intrathecal administration of Poly (I:C), a TLR3 agonist, significantly increases the activation of microglial autophagy, whereas TLR3 knockdown markedly inhibits L5 SNL-induced microglial autophagy. Poly (I:C) treatment promotes the expression of proinflammatory mediators, whereas 3-MA (a specific inhibitor of autophagy) suppresses Poly (I:C)-induced secretion of proinflammatory cytokines. Autophagy inhibition further inhibits TLR3-mediated mechanical and cold hypersensitivity following SNL. These results suggest that inhibition of TLR3/autophagy signaling contributes to alleviate neurophathic pain triggered by SNL.





Similar content being viewed by others
References
Ellis A, Bennett DL (2013) Neuroinflammation and the generation of neuropathic pain. Br J Anaesth 111:26–37
Masuda T, Iwamoto S, Yoshinaga R, Tozaki-Saitoh H, Nishiyama A, Mak TW, Tamura T, Tsuda M, Inoue K (2014) Transcription factor IRF5 drives P2 × 4R+-reactive microglia gating neuropathic pain. Nat Commun 5:3771
Myers RR, Campana WM, Shubayev VI (2006) The role of neuroinflammation in neuropathic pain: mechanisms and therapeutic targets. Drug Discov Today 11:8–20
Takeuchi O, Akira S (2010) Pattern recognition receptors and inflammation. Cell 140:805–820
Nguyen MD, Julien JP, Rivest S (2002) Innate immunity: the missing link in neuroprotection and neurodegeneration? Nat Rev Neurosci 3:216–227
Tanga FY, Nutile-McMenemy N, DeLeo JA (2005) The CNS role of Toll-like receptor 4 in innate neuroimmunity and painful neuropathy. Proc Natl Acad Sci USA 102:5856–5861
Mei XP, Zhou Y, Wang W, Tang J, Zhang H, Xu LX, Li YQ (2011) Ketamine depresses toll-like receptor 3 signaling in spinal microglia in a rat model of neuropathic pain. Neurosignals 19:44–53
Li X, Cudaback E, Keene CD, Breyer RM, Montine TJ (2011) Suppressed microglial E prostanoid receptor 1 signaling selectively reduces tumor necrosis factor alpha and interleukin 6 secretion from toll-like receptor 3 activation. Glia 59:569–576
Bettoni I, Comelli F, Rossini C, Granucci F, Giagnoni G, Peri F, Costa B (2008) Glial TLR4 receptor as new target to treat neuropathic pain: efficacy of a new receptor antagonist in a model of peripheral nerve injury in mice. Glia 56:1312–1319
Kim D, Kim MA, Cho IH, Kim MS, Lee S, Jo EK, Choi SY, Park K, Kim JS, Akira S, Na HS, Oh SB, Lee SJ (2007) A critical role of toll-like receptor 2 in nerve injury-induced spinal cord glial cell activation and pain hypersensitivity. J Biol Chem 282:14975–14983
Lim H, Kim D, Lee SJ (2013) Toll-like receptor 2 mediates peripheral nerve injury-induced NADPH oxidase 2 expression in spinal cord microglia. J Biol Chem 288:7572–7579
Arroyo DS, Soria JA, Gaviglio EA, Garcia-Keller C, Cancela LM, Rodriguez-Galan MC, Wang JM, Iribarren P (2013) Toll-like receptor 2 ligands promote microglial cell death by inducing autophagy. FASEB J 27:299–312
Yang Z, Liu B, Zhong L, Shen H, Lin C, Lin L, Zhang N, Yuan B (2015) Toll-like receptor-4-mediated autophagy contributes to microglial activation and inflammatory injury in mouse models of intracerebral haemorrhage. Neuropathol Appl Neurobiol 41:e95–e106
Mei XP, Zhou Y, Wang W, Tang J, Wang W, Zhang H, Xu LX, Li YQ (2011) Ketamine depresses toll-like receptor 3 signaling in spinal microglia in a rat model of neuropathic pain. Neurosignals 19:44–53
Obata K, Katsura H, Miyoshi K, Kondo T, Yamanaka H, Kobayashi K, Dai Y, Fukuoka T, Akira S, Noguchi K (2008) Toll-like receptor 3 contributes to spinal glial activation and tactile allodynia after nerve injury. J Neurochem 105:2249–2259
Klionsky DJ, Emr SD (2000) Autophagy as a regulated pathway of cellular degradation. Science 290:1717–1721
Srivastava IN, Shperdheja J, Baybis M, Ferguson T, Crino PB (2015) mTOR pathway inhibition prevents neuroinflammation and neuronal death in a mouse model of cerebral palsy. Neurobiol Dis 85:144–154
Shi G, Shi J, Liu K, Liu N, Wang Y, Fu Z, Ding J, Jia L, Yuan W (2013) Increased miR-195 aggravates neuropathic pain by inhibiting autophagy following peripheral nerve injury. Glia 61:504–512
Kim SH, Chung JM (1992) An experimental model for peripheral neuropathy produced by segmental spinal nerve ligation in the rat. Pain 50:355–363
Ge Y, Wu F, Sun X, Xiang Z, Yang L, Huang S, Lu Z, Sun Y, Yu WF (2014) Intrathecal infusion of hydrogen-rich normal saline attenuates neuropathic pain via inhibition of activation of spinal astrocytes and microglia in rats. PLoS One 9:e97436
Ueno S, Yamada H, Moriyama T, Honda K, Takano Y, Kamiya HO, Katsuragi T (2002) Measurement of dorsal root ganglion P2X mRNA by SYBR Green fluorescence. Brain Res Brain Res Protoc 10:95–101
Dominguez E, Rivat C, Pommier B, Mauborgne A, Pohl M (2008) JAK/STAT3 pathway is activated in spinal cord microglia after peripheral nerve injury and contributes to neuropathic pain development in rat. J Neurochem 107:50–60
Calvo M, Zhu N, Grist J, Ma Z, Loeb JA, Bennett DL (2011) Following nerve injury neuregulin-1 drives microglial proliferation and neuropathic pain via the MEK/ERK pathway. Glia 59:554–568
Khorooshi R, Morch MT, Holm TH, Berg CT, Dieu RT, Draeby D, Issazadeh-Navikas S, Weiss S, Lienenklaus S, Owens T (2015) Induction of endogenous Type I interferon within the central nervous system plays a protective role in experimental autoimmune encephalomyelitis. Acta Neuropathol (Berl) 130:107–118
Stokes JA, Corr M, Yaksh TL (2013) Spinal toll-like receptor signaling and nociceptive processing: regulatory balance between TIRAP and TRIF cascades mediated by TNF and IFNbeta. Pain 154:733–742
Klionsky DJ, Abeliovich H, Agostinis P, Agrawal DK, Aliev G, Askew DS et al (2008) Guidelines for the use and interpretation of assays for monitoring autophagy in higher eukaryotes. Autophagy 4:151–175
Berliocchi L, Russo R, Maiaru M, Levato A, Bagetta G, Corasaniti MT (2011) Autophagy impairment in a mouse model of neuropathic pain. Mol Pain 7:83
Bowman CC, Rasley A, Tranguch SL, Marriott I (2003) Cultured astrocytes express toll-like receptors for bacterial products. Glia 43:281–291
Jack CS, Arbour N, Manusow J, Montgrain V, Blain M, McCrea E, Shapiro A, Antel JP (2005) TLR signaling tailors innate immune responses in human microglia and astrocytes. J Immunol 175:4320–4330
Qi J, Buzas K, Fan H, Cohen JI, Wang K, Mont E, Klinman D, Oppenheim JJ, Howard OM (2011) Painful pathways induced by TLR stimulation of dorsal root ganglion neurons. J Immunol 186:6417–6426
Bsibsi M, Ravid R, Gveric D, van Noort JM (2002) Broad expression of Toll-like receptors in the human central nervous system. J Neuropathol Exp Neurol 61:1013–1021
Levine B, Klionsky DJ (2004) Development by self-digestion: molecular mechanisms and biological functions of autophagy. Dev Cell 6:463–477
Suzuki Y, Maazi H, Sankaranarayanan I, Lam J, Khoo B, Soroosh P, Barbers RG, James Ou JH, Jung JU, Akbari O (2015) Lack of autophagy induces steroid-resistant airway inflammation. J Allergy Clin Immunol 137(5):1382–1389
Rufini S, Ciccacci C, Di Fusco D, Ruffa A, Pallone F, Novelli G, Biancone L, Borgiani P (2015) Autophagy and inflammatory bowel disease: Association between variants of the autophagy-related IRGM gene and susceptibility to Crohn’s disease. Dig Liver Dis 47:744–750
Motori E, Puyal J, Toni N, Ghanem A, Angeloni C, Malaguti M, Cantelli-Forti G, Berninger B, Conzelmann KK, Gotz M, Winklhofer KF, Hrelia S, Bergami M (2013) Inflammation-induced alteration of astrocyte mitochondrial dynamics requires autophagy for mitochondrial network maintenance. Cell Metab 18:844–859
Liang P, Le W (2015) Role of autophagy in the pathogenesis of multiple sclerosis. Neurosci Bull 31:435–444
Shi CS, Shenderov K, Huang NN, Kabat J, Abu-Asab M, Fitzgerald KA, Sher A, Kehrl JH (2012) Activation of autophagy by inflammatory signals limits IL-1beta production by targeting ubiquitinated inflammasomes for destruction. Nat Immunol 13:255–263
Deretic V, Saitoh T, Akira S (2013) Autophagy in infection, inflammation and immunity. Nat Rev Immunol 13:722–737
Saitoh T, Fujita N, Jang MH, Uematsu S, Yang BG, Satoh T, Omori H, Noda T, Yamamoto N, Komatsu M, Tanaka K, Kawai T, Tsujimura T, Takeuchi O, Yoshimori T, Akira S (2008) Loss of the autophagy protein Atg16L1 enhances endotoxin-induced IL-1beta production. Nature 456:264–268
Guo ML, Liao K, Periyasamy P, Yang L, Cai Y, Callen SE, Buch S (2015) Cocaine-mediated microglial activation involves the ER stress-autophagy axis. Autophagy 11:995–1009
Xu Y, Jagannath C, Liu XD, Sharafkhaneh A, Kolodziejska KE, Eissa NT (2007) Toll-like receptor 4 is a sensor for autophagy associated with innate immunity. Immunity 27:135–144
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest exist.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, W., Lu, Z. Upregulated TLR3 Promotes Neuropathic Pain by Regulating Autophagy in Rat With L5 Spinal Nerve Ligation Model. Neurochem Res 42, 634–643 (2017). https://doi.org/10.1007/s11064-016-2119-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-016-2119-2