Abstract
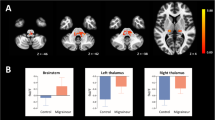
The resting state amplitude of low-frequency fluctuations (ALFF) in functional magnetic resonance imaging (fMRI) is believed to reflect spontaneous cerebral neural activity. The pathophysiology of blepharospasm (BSP), which is characterized by motor symptoms and also sensory symptoms, remains unclear. The present study aims to localize possible cerebral functional abnormalities in BSP patients using resting state fMRI, and explore their possible associations with clinical variables. Voxel-based analysis was used to characterize the difference of ALFF between eighteen BSP patients and eighteen matched healthy controls. Separate correlation analyses were conducted to explore the possible association between ALFF values of significantly different areas and clinical measures including onset age, disease duration, symptom severity evaluated by Jankovic rating scale (JRS), and presence of geste antagoniste. The whole-brain analysis indicated that the BSP group had significantly decreased ALFF in the left thalamus while increased ALFF in the left orbitofrontal areas extending from middle frontal gyrus to inferior frontal gyrus. The mean ALFF in the left thalamus was negatively correlated with the subscore of JRS-frequency (r = −0.484, p = 0.042) and JRS total score (r = −0.477, p = 0.045). A borderline positive correlation was detected between the mean ALFF in the left orbitofrontal area and disease duration(r = 0.485, p = 0.049). Our findings suggest sensorimotor integration is abnormal in BSP, and dysfunctional activity of thalamus may be used to measuring symptom severity in BSP patients.


Similar content being viewed by others
References
Hallett M, Evinger C, Jankovic J, Stacy M (2008) Update on blepharospasm: report from the BEBRF international workshop. Neurology 71(16):1275–1282. doi:10.1212/01.wnl.0000327601.46315.85
Herz NL, Yen MT (2005) Modulation of sensory photophobia in essential blepharospasm with chromatic lenses. Ophthalmology 112(12):2208–2211. doi:10.1016/j.ophtha.2005.06.030
Hallett M (1995) Is dystonia a sensory disorder? Ann Neurol 38(2):139–140. doi:10.1002/ana.410380203
Baker RS, Andersen AH, Morecraft RJ, Smith CD (2003) A functional magnetic resonance imaging study in patients with benign essential blepharospasm. J Neuroophthalmol 23(1):11–15
Dresel C, Haslinger B, Castrop F, Wohlschlaeger AM, Ceballos-Baumann AO (2006) Silent event-related fMRI reveals deficient motor and enhanced somatosensory activation in orofacial dystonia. Brain 129(Pt 1):36–46. doi:10.1093/brain/awh665
Goncalves SI, de Munck JC, Pouwels PJ, Schoonhoven R, Kuijer JP, Maurits NM, Hoogduin JM, Van Someren EJ, Heethaar RM, Lopes da Silva FH (2006) Correlating the alpha rhythm to BOLD using simultaneous EEG/fMRI: inter-subject variability. Neuroimage 30(1):203–213. doi:10.1016/j.neuroimage.2005.09.062
Zang YF, He Y, Zhu CZ, Cao QJ, Sui MQ, Liang M, Tian LX, Jiang TZ, Wang YF (2007) Altered baseline brain activity in children with ADHD revealed by resting-state functional MRI. Brain Dev 29(2):83–91. doi:10.1016/j.braindev.2006.07.002
Luo C, Chen Q, Huang R, Chen X, Chen K, Huang X, Tang H, Gong Q, Shang HF (2012) Patterns of spontaneous brain activity in amyotrophic lateral sclerosis: a resting-state FMRI study. PLoS One 7(9):e45470. doi:10.1371/journal.pone.0045470
Guo WB, Liu F, Xun GL, Hu MR, Guo XF, Xiao CQ, Chen HF, Wooderson SC, Chen JD, Zhao JP (2013) Reversal alterations of amplitude of low-frequency fluctuations in early and late onset, first-episode, drug-naive depression. Prog Neuropsychopharmacol Biol Psychia 40:153–159. doi:10.1016/j.pnpbp.2012.08.014
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9(1):97–113
Fahn S, Marsden CD, Calne DB (1987) Classification and investigation of dystonia. In: Marsden CD, Fahn S (eds) Movement disorders, vol 2. Butterworth, London, pp 332–358
Jankovic J, Orman J (1987) Botulinum A toxin for cranial-cervical dystonia: a double-blind, placebo-controlled study. Neurology 37(4):616–623
Oakes TR, Fox AS, Johnstone T, Chung MK, Kalin N, Davidson RJ (2007) Integrating VBM into the general linear model with voxel wise anatomical covariates. Neuroimage 34(2):500–508. doi:10.1016/j.neuroimage.2006.10.007
Ashburner J (2007) A fast diffeomorphic image registration algorithm. Neuroimage 38(1):95–113. doi:10.1016/j.neuroimage.2007.07.007
Zoons E, Booij J, Nederveen AJ, Dijk JM, Tijssen MAJ (2011) Structural, functional and molecular imaging of the brain in primary focal dystonia—a review. Neuroimage 56(3):1011–1020. doi:10.1016/j.neuroimage.2011.02.045
Obermann M, Yaldizli O, De Greiff A, Lachenmayer ML, Buhl AR, Tumczak F, Gizewski ER, Diener HC, Maschke M (2007) Morphometric changes of sensorimotor structures in focal dystonia. Mov Disord 22(8):1117–1123. doi:10.1002/mds.21495
Miranda M, Millar A (1998) Blepharospasm associated with bilateral infarcts confined to the thalamus: case report. Mov Disord 13(3):616–617. doi:10.1002/mds.870130347
Behrens TE, Johansen-Berg H, Woolrich MW, Smith SM, Wheeler-Kingshott CA, Boulby PA, Barker GJ, Sillery EL, Sheehan K, Ciccarelli O, Thompson AJ, Brady JM, Matthews PM (2003) Non-invasive mapping of connections between human thalamus and cortex using diffusion imaging. Nat Neurosci 6(7):750–757. doi:10.1038/nn1075
Bollen E, Van Exel E, van der Velde EA, Buytels P, Bastiaanse J, van Dijk JG (1996) Saccadic eye movements in idiopathic blepharospasm. Mov Disord 11(6):678–682. doi:10.1002/mds.870110613
Arend I, Machado L, Ward R, McGrath M, Ro T, Rafal RD (2008) The role of the human pulvinar in visual attention and action: evidence from temporal-order judgment, saccade decision, and anti saccade tasks. Prog Brain Res 171:475–483. doi:10.1016/s0079-6123(08)00669-9
Emoto H, Suzuki Y, Wakakura M, Horie C, Kiyosawa M, Mochizuki M, Kawasaki K, Oda K, Ishiwata K, Ishii K (2010) Photophobia in essential blepharospasm––a positron emission tomographic study. Mov Disord 25(4):433–439. doi:10.1002/mds.22916
Scott RB, Gregory R, Wilson J, Banks S, Turner A, Parkin S, Giladi N, Joint C, Aziz T (2003) Executive cognitive deficits in primary dystonia. Mov Disord 18(5):539–550. doi:10.1002/mds.10399
Hinse P, Leplow B, Humbert T, Lamparter U, Junge A, Emskotter T (1996) Impairment of visuospatial function in idiopathic spasmodic torticollis. J Neurol 243(1):29–33
Saalmann YB, Pinsk MA, Wang L, Li X, Kastner S (2012) The pulvinar regulates information transmission between cortical areas based on attention demands. Science 337(6095):753–756. doi:10.1126/science.1223082
Danziger S, Ward R, Owen V, Rafal R (2004) Contributions of the human pulvinar to linking vision and action. Cogn Affect Behav Neurosci 4(1):89–99
Kerrison JB, Lancaster JL, Zamarripa FE, Richardson LA, Morrison JC, Holck DE, Andreason KW, Blaydon SM, Fox PT (2003) Positron emission tomography scanning in essential blepharospasm. Am J Ophthalmol 136(5):846–852
Tsubota K, Kwong KK, Lee TY, Nakamura J, Cheng HM (1999) Functional MRI of brain activation by eye blinking. Exp Eye Res 69(1):1–7. doi:10.1006/exer.1999.0660
Swick D, Ashley V, Turken AU (2008) Left inferior frontal gyrus is critical for response inhibition. BMC Neurosci 9(1):102. doi:10.1186/1471-2202-9-102
Simonyan K, Ludlow CL (2010) Abnormal activation of the primary somatosensory cortex in spasmodic dysphonia: an fMRI study. Cereb Cortex 20(11):2749–2759. doi:10.1093/cercor/bhq023
Egger K, Mueller J, Schocke M, Brenneis C, Rinnerthaler M, Seppi K, Trieb T, Wenning GK, Hallett M, Poewe W (2007) Voxel based morphometry reveals specific gray matter changes in primary dystonia. Mov Disord 22(11):1538–1542. doi:10.1002/mds.21619
Drevets WC (2007) Orbitofrontal cortex function and structure in depression. Ann N Y Acad Sci 1121:499–527. doi:10.1196/annals.1401.029
Lagemann T, Rentzsch J, Montag C, Gallinat J, Jockers-Scherubl M, Winter C, Reischies FM (2012) Early orbitofrontal hyper activation in obsessive-compulsive disorder. Psychiatry Res 202(3):257–263. doi:10.1016/j.pscychresns.2011.10.002
Broocks A, Thiel A, Angerstein D, Dressler D (1998) Higher prevalence of obsessive-compulsive symptoms in patients with blepharospasm than in patients with hemifacial spasm. Am J Psychia 155(4):555–557
Lencer R, Steinlechner S, Stahlberg J, Rehling H, Orth M, Baeumer T, Rumpf HJ, Meyer C, Klein C, Muenchau A, Hagenah J (2009) Primary focal dystonia: evidence for distinct neuropsychiatric and personality profiles. J Neurol Neurosurg Psychiatry 80(10):1176–1179. doi:10.1136/jnnp.2008.170191
Zhou B, Wang J, Huang Y, Yang Y, Gong Q, Zhou D (2013) A resting state functional magnetic resonance imaging study of patients with benign essential blepharospasm. J Neuroophthalmol. doi:10.1097/WNO.0b013e31828f69e5
Acknowledgments
This study was supported by the National Natural Science Foundation (Grant Nos. 30973149, 81030027, 81227002 and 81220108013) and National Key Technologies R&D Program (Program No. 2012BAI01B03) of China. We thank Ms. XiaoYan Guo for statistical assistance, and thank the technical staff of the Department of Radiology for their collaboration and assistance.
Conflicts of interest
No conflict of interest concerning the research related to the manuscript and no founding support.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Yang, J., Luo, C., Song, W. et al. Altered regional spontaneous neuronal activity in blepharospasm: a resting state fMRI study. J Neurol 260, 2754–2760 (2013). https://doi.org/10.1007/s00415-013-7042-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-013-7042-8