Abstract
Main conclusion
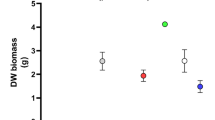
Microalgae treated with blue light have potential for production of human nutrition supplement and biofuel due to their higher biomass productivity and favorable fatty acid composition.
Abstract
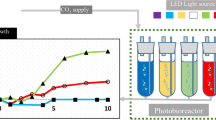
Chlorella vulgaris, Chlorella pyrenoidosa, Scenedesmus quadricauda and Scenedesmus obliquus are representative green microalgae which are widely reported for algal production. In this study, we provide a systematic investigation of the biomass productivity, photosynthetic pigments, chlorophyll fluorescence and fatty acid content of the four green microalgae. The strains were grown in two primary monochromatic light wavelengths [red and blue LEDs (light emitting diode)], and in white LED conditions, respectively. Among them, blue LED light was determined as the best light for growth rate, followed by red LED and white LED. The chlorophyll generation was more sensitive to the monochromatic blue light. The polyunsaturated fatty acids (PUFAs) such as α-linolenic acid (18:3), which were perfect for human nutrition supplementation, showed high concentrations in these algae strains under blue LED. Collectively, the results indicate that the blue LED is suitable for various food, feed, and algal biofuel productions due to both biomass and fatty acid productivity.



Similar content being viewed by others
Abbreviations
- ABS:
-
Absorption
- ET0 :
-
Electron transport
- DI0 :
-
Dissipation
- RC:
-
Reaction center
- TR0 :
-
Trapping
References
Atta M, Idris A, Bukhari A, Wahidin S (2013) Intensity of blue LED light: a potential stimulus for biomass and lipid content in fresh water microalgae Chlorella vulgaris. Bioresour Technol 148:373–378
Cerón MC, García-Malea MC, Rivas J, Acién FG, Fernández JM, Del Río E, Guerrero MG, Molina E (2007) Antioxidant activity of Haematococcus pluvialis cells grown in continuous culture as a function of their carotenoid and fatty acid content. Appl Microbiol Biotechnol 274:1112–1119
Chacon-Lee TL, Gonzalez-Marino GE (2010) Microalgae for “healthy” foods-possibilities and challenges. Compr Rev Food Sci Food Saf 9:655–675
Dahmen I, Chtourou H, Jebali A, Daassi D, Karray F, Hassairi I, Sayadi S, Abdelkafi S, Dhouib A (2013) Optimisation of the critical medium components for better growth of Picochlorum sp. and the role of stressful environments for higher lipid production. J Sci Food Agric 94:1628–1638
Das P, Wang L, Aziz SS, Obbard JP (2011) Enhanced algae growth in both phototrophic and mixotrophic culture under blue light. Bioresour Technol 102:3883–3887
Del Campo JA, Moreno J, Rodríguez H, Angeles Vargas M, Rivas J, Guerrero MG (2000) Carotenoid content of chlorophycean microalgae: factors determining lutein accumulation in Muriellopsis sp. (Chlorophyta). J Biotechnol 76:51–59
Dubinsky Z, Matsukawa R, Karube I (1995) Photobiological aspects of algal mass cultures. J Mar Biotechnol 2:61–65
FAO (2010) Fats and fatty acids in human nutrition. Report of an expert consultation. FAO Food and nutrition 91
Kebede E, Ahlgren G (1996) Optimum growth conditions and light utilization efficiency of Spirulina platensis (= Arthrospira fusiformis) (Cyanophyta) from Lake Chitu, Ethiopia. Hydrobiologia 332:99–109
Kuo F, Chien Y, Chen C (2012) Effect of light sources on growth and carotenoid content of photosynthetic bacterial Rhodopseudomonas palustris. Bioresour Technol 113:315–318
Ma G, Zhang L, Kato M, Yamawaki K, Kiriiwa Y, Yahata M, Ikoma Y, Matsumoto H (2012) Effect of blue and red LED light irradiation on β-cryptoxanthin accumulation in the flavedo of citrus fruits. J Agric Food Chem 60:197–201
Mahapatra DM, Chanakya HN, Ramachandra TV (2013) Euglena ap. As a suitable source of lipids for potential use as biofuel and sustainable wastewater treatment. J Appl Phycol 25:855–865
Mata TM, Martins AA, Caetano NS (2010) Microalgae for biodiesel production and other applications: a review. Renew Sust Energy Rev 14:217–232
Matthijs HC, Balke H, Van Hes UM, Kroon B, Mur LR, Binot RA (1996) Application of light-emitting diodes in bioreactors: flashing light effects and energy economy in algal culture (Chlorella pyrenoidosa). Biotechnol Bioeng 50:98–107
Michael C, Del Ninno M, Gross M, Wen Z (2015) Use of wavelength-selective optical light filters for enhanced microalgal growth in different algal cultivation systems. Bioresour Technol 179:473–482
Minguez-Mosquera MI, Gandul-Rojas B, Gallardo-Guerrero ML (1992) Rapid method of quantification of chlorophylls and carotenoids in virgin olive oil by high-performance liquid chromatography. J Agric Food Chem 40:60–63
Mouget JL, Rosa P, Vachoux C, Tremblin G (2005) Enhancement of marennine production by blue light in the diatom Haslea ostrearia. J Appl Phycol 17:437–445
Nussbaum S, Geissmann M, Eggenberg P, Strasser RJ, Fuhrer J (2001) Ozone sensitivity in herbaceous species as assessed by direct and modulated chlorophyll fluorescence techniques. J Plant Physiol 158:757–766
Rodolfi L, Zittelli GC, Bassi N, Padovani G, Biondi N, Bonini G, Tredici MR (2009) Microalgae for oil: strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol Bioeng 102:100–112
Rosenberg JN, Oyler GA, Wilkinson L, Betenbaugh MJ (2008) A green light for engineered algae: redirecting metabolism to fuel a biotechnology revolution. Curr Opin Biotech 19:430–436
Ruiz-Vázquez R, Fontes M, Murillo FJ (1993) Clustering and coordinated activation of carotenoid genes in Myxococcus xanthus by blue light. Mol Microbiol 10:25–34
Ruyters G (1984) Effects of blue light on enzymes. In: Senger H (ed) Blue light effects in biological systems. Springer, Berlin, Heidelberg, pp 283–301
Sánchez-Saavedra MP, Voltolina D (2006) The growth rate, biomass production and composition of Chaetoceros sp. grown with different light sources. Aquacult Eng 35:161–165
Shu CH, Tsai CC, Liao WH, Chen KY, Huang HC (2012) Effects of light quality on the accumulation of oil in a mixed culture of Chlorella sp. and Saccharomyces cerevisae. J Chem Technol Biot 87:601–607
Simoupolos AP (2006) Evolutionary aspects of diet, the omega-6/omega-3 ratio and genetic variation: nutritional implications for chronic disease. Biomed Pharmacother 60:502–507
Strasser BJ, Strasser RJ (1995) Measuring fast fluorescence transients to address environmental questions: the JIP test. In: Mathis P (ed) Photosynthesis: from light to biosphere, vol 5. Kluwer Academic Publisher, The Netherlands, pp 977–980
Tamburic B, Szabó M, Tran NA, Larkum AWD, Suggett DJ (2014) Action spectra of oxygen production and chlorophyll a fluorescence in the green microalga Nannochloropsis oculata. Bioresour Technol 169:320–327
Teo CL, Jamaluddin H, Zain NAM, Idris A (2014) Biodiesel production via lipase catalysed transesterification of microalgae lipids from Tetraselmis sp. Renew Energy 68:1–5
Vadiveloo A, Moheimani NR, Cosgrove JJ, Bahri PA, Parlevliet D (2015) Effect of different light spectra on the growth and productivity of acclimated Nannochloropsis sp. (Eustigmatophyceae). Algal Res 8:121–127
Voskresenskaya NP (1972) Blue light and carbon metabolism. Annu Rev Plant Physiol 23:219–234
Wang CY, Fu CC, Liu YC (2007) Effects of using light-emitting diodes on the cultivation of Spirulina platensis. Biochem Eng J 37:21–25
Zhu XC, Song FB, Xu HW (2010) Arbuscular mycorrhizae improves low temperature stress in maize via alterations in host water status and photosynthesis. Plant Soil 331:129–137
Acknowledgements
This research was mainly funded by a Key Project of Shenzhen Emerging Industries (No. JC201104210118A) and partially supported by Public Science and Technology Research Funds Projects of Ocean (201305022).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhong, Y., Jin, P. & Cheng, J.J. A comprehensive comparable study of the physiological properties of four microalgal species under different light wavelength conditions. Planta 248, 489–498 (2018). https://doi.org/10.1007/s00425-018-2899-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-018-2899-5