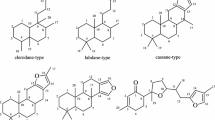
Phytochemical studies on the EtOH extract of Drypetes hainanensis leaves and stems afforded a new friedelane-type triterpene named 4β-hydroxy-23-nor-friedel-3-one (1), along with seven known compounds: friedelin (2), diethylhexyl phthalate (3), α-amyrin (4), β-amyrin (5), lupeol (6), friedelane-3,7-dione (7) and β-sitosterol (8). Their structures were established on the basis of spectroscopic analysis and chemical evidence. The cytotoxicity of the selected compounds 1, 2, and 7 were evaluated in vitro against three human cancer cell lines (HL-60, BEL-7402, and A549). Compounds 1 and 7 showed moderate cytotoxic activity.

Similar content being viewed by others
References
Bing Tao Li, Flora of China, Vol. 44, Science Press, Beijing, 1994.
A. Bouquet and L. Debray, Plantes Medicinales de la Cote-d’Ivoire, 32, Travaux et documents de l’ORSTOM, Paris, 1974, 82 pp.
R. F. Irvine, Woody Plants of Ghana, Oxford University Press, London, 1961, 223 pp.
A. R. Walker, R. Sillans, and J. L. Trochain, Les Plantes Utiles du Gabon, P. Lechevalier (ed.), 12-Rue de Tournon, Paris VI, Paris, 1961, 165 pp.
Chen De-li, Xiao Man, and Liu Ping-huai, Fine Chem., 11, 1103 (2011).
Margaret Mwihaki Ng’ang’a, Hidayat Hussain, Sumesh Chhabra, Caroline Langat-Thoruwa, Beatrice N. Irungu, Ahmed Al-Harrasi, Muhammad Riaz, and Karsten Krohn, Chem. Nat. Compd., 48, 339 (2012).
S. B. Mahato and A. P. Kundu, Phytochemistry, 37, 1517 (1994).
J. C. Lhuguenot, Peroxisomes: Biology and Importance in Toxicology and Medicine, Taylor and Francis, London, 1993, 4651 pp.
X. Li, Y. H. Yao, and Y. N. Zheng, Chin. J. Nat. Med., 13, 188 (2006).
R. S. Gallegos and N. E. Roque, Quimica. Nova, 4, 278 (1990).
S. B. Mahato and S. Sen, Phytochemistry, 44 (7), 1185 (1997).
J. Wanji, F. Tillequin, D. A. Mulholland, A. D. Temgoua, J. D. Wansi, E. Seguin, and Z. T. Fomum, Phytochemistry, 63, 453 (2003).
N. B. Chungag-Anye, D. Njamen, J. Wandji, Z. T. Fomum, A. Dongmo, T. B. Nguelefack, D. Wansi, and A. Kamanyi, Pharm. Biol., 1, 26 (2003).
T. Mosmman, J. Immunol. Methods, 1–2, 55 (1983).
Vanicha Vichai and Kanyawim Kirtikara, Nat. Protocols, 3, 1112 (2006).
P. Skehan, P. Storeng, D. Scudiero, A. Monks, J. McMahon, D. Vistica, J. T. Warren, H. Bokesch, S. Kenney, and M. R. Boyd, J. Natl. Cancer Inst., 82, 1107 (1990).
Acknowledgment
The authors acknowledge the technical assistance of the Analytical and Testing Center of Hainan University. This article is supported by the National Natural Science Foundation of Hainan (Grant No. 309001) and the State key subject of Botany at Hainan University (071001).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 1, January–February, 2014, pp. 84–86.
Rights and permissions
About this article
Cite this article
Chen, D., Cheng, X., Sun, Y. et al. A New Friedelane Triterpenoid Possessing Cytotoxicity from the Leaves and Stems OF Drypetes hainanensis . Chem Nat Compd 50, 93–96 (2014). https://doi.org/10.1007/s10600-014-0874-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-014-0874-3