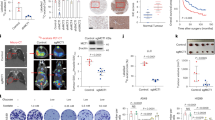
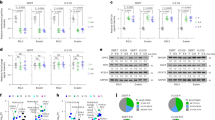
Abstract
Metabolic reprograming is an emerging hallmark of tumor biology and an actively pursued opportunity in discovery of oncology drugs. Extensive efforts have focused on therapeutic targeting of glycolysis, whereas drugging mitochondrial oxidative phosphorylation (OXPHOS) has remained largely unexplored, partly owing to an incomplete understanding of tumor contexts in which OXPHOS is essential. Here, we report the discovery of IACS-010759, a clinical-grade small-molecule inhibitor of complex I of the mitochondrial electron transport chain. Treatment with IACS-010759 robustly inhibited proliferation and induced apoptosis in models of brain cancer and acute myeloid leukemia (AML) reliant on OXPHOS, likely owing to a combination of energy depletion and reduced aspartate production that leads to impaired nucleotide biosynthesis. In models of brain cancer and AML, tumor growth was potently inhibited in vivo following IACS-010759 treatment at well-tolerated doses. IACS-010759 is currently being evaluated in phase 1 clinical trials in relapsed/refractory AML and solid tumors.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Warburg, O., Wind, F. & Negelein, E. The metabolism of tumors in the body. J. Gen. Physiol. 8, 519–530 (1927).
Palaskas, N. et al. 18F-fluorodeoxy-glucose positron emission tomography marks MYC-overexpressing human basal-like breast cancers. Cancer Res. 71, 5164–5174 (2011).
DeBerardinis, R. J. & Chandel, N. S. Fundamentals of cancer metabolism. Sci. Adv. 2, e1600200 (2016).
Koppenol, W. H., Bounds, P. L. & Dang, C. V. Otto Warburg’s contributions to current concepts of cancer metabolism. Nat. Rev. Cancer 11, 325–337 (2011).
Caro, P. et al. Metabolic signatures uncover distinct targets in molecular subsets of diffuse large B cell lymphoma. Cancer Cell 22, 547–560 (2012).
Goto, M. et al. Importance of glutamine metabolism in leukemia cells by energy production through TCA cycle and by redox homeostasis. Cancer Invest. 32, 241–247 (2014).
Goto, M. et al. Adaptation of leukemia cells to hypoxic condition through switching the energy metabolism or avoiding the oxidative stress. BMC Cancer 14, 76 (2014).
Haq, R. et al. Oncogenic BRAF regulates oxidative metabolism via PGC1α and MITF. Cancer Cell 23, 302–315 (2013).
Hu, J. et al. Heterogeneity of tumor-induced gene expression changes in the human metabolic network. Nat. Biotechnol. 31, 522–529 (2013).
Roesch, A. et al. Overcoming intrinsic multidrug resistance in melanoma by blocking the mitochondrial respiratory chain of slow-cycling JARID1Bhigh cells. Cancer Cell 23, 811–825 (2013).
Simsek, T. et al. The distinct metabolic profile of hematopoietic stem cells reflects their location in a hypoxic niche. Cell Stem Cell 7, 380–390 (2010).
Skrtić, M. et al. Inhibition of mitochondrial translation as a therapeutic strategy for human acute myeloid leukemia. Cancer Cell 20, 674–688 (2011).
Sriskanthadevan, S. et al. AML cells have low spare reserve capacity in their respiratory chain that renders them susceptible to oxidative metabolic stress. Blood 125, 2120–2130 (2015).
Vazquez, F. et al. PGC1α expression defines a subset of human melanoma tumors with increased mitochondrial capacity and resistance to oxidative stress. Cancer Cell 23, 287–301 (2013).
Viale, A. et al. Oncogene ablation–resistant pancreatic cancer cells depend on mitochondrial function. Nature 514, 628–632 (2014).
Birsoy, K. et al. An essential role of the mitochondrial electron transport chain in cell proliferation is to enable aspartate synthesis. Cell 162, 540–551 (2015).
Sullivan, L. B. et al. Supporting aspartate biosynthesis is an essential function of respiration in proliferating cells. Cell 162, 552–563 (2015).
Bridges, H. R., Jones, A. J., Pollak, M. N. & Hirst, J. Effects of metformin and other biguanides on oxidative phosphorylation in mitochondria. Biochem. J. 462, 475–487 (2014).
Dykens, J. A. et al. Biguanide-induced mitochondrial dysfunction yields increased lactate production and cytotoxicity of aerobically-poised HepG2 cells and human hepatocytes in vitro. Toxicol. Appl. Pharmacol. 233, 203–210 (2008).
Wang, D. S. et al. Involvement of organic cation transporter 1 in hepatic and intestinal distribution of metformin. J. Pharmacol. Exp. Ther. 302, 510–515 (2002).
Sanchez, M., Gastaldi, L., Remedi, M., Cáceres, A. & Landa, C. Rotenone-induced toxicity is mediated by Rho-GTPases in hippocampal neurons. Toxicol. Sci. 104, 352–361 (2008).
Ellinghaus, P. et al. BAY 87-2243, a highly potent and selective inhibitor of hypoxia-induced gene activation has antitumor activities by inhibition of mitochondrial complex I. Cancer Med. 2, 611–624 (2013).
Li, S. H. et al. A novel mode of action of YC-1 in HIF inhibition: stimulation of FIH-dependent p300 dissociation from HIF-1α. Mol. Cancer Ther. 7, 3729–3738 (2008).
Lin, X. et al. A chemical genomics screen highlights the essential role of mitochondria in HIF-1 regulation. Proc. Natl. Acad. Sci. USA 105, 174–179 (2008).
Bai, Y. et al. Lack of complex I activity in human cells carrying a mutation in MtDNA-encoded ND4 subunit is corrected by the Saccharomyces cerevisiae NADH-quinone oxidoreductase (NDI1) gene. J. Biol. Chem. 276, 38808–38813 (2001).
Seo, B. B. et al. Molecular remedy of complex I defects: rotenone-insensitive internal NADH-quinone oxidoreductase of Saccharomyces cerevisiae mitochondria restores the NADH oxidase activity of complex I-deficient mammalian cells. Proc. Natl Acad. Sci. USA 95, 9167–9171 (1998).
Petrova-Benedict, R., Buncic, J. R., Wallace, D. C. & Robinson, B. H. Selective killing of cells with oxidative defects in galactose medium: a screening test for affected patient fibroblasts. J. Inherit. Metab. Dis. 15, 943–944 (1992).
Robinson, B. H., Petrova-Benedict, R., Buncic, J. R. & Wallace, D. C. Nonviability of cells with oxidative defects in galactose medium: a screening test for affected patient fibroblasts. Biochem. Med. Metab. Biol. 48, 122–126 (1992).
Zhu, J., Vinothkumar, K. R. & Hirst, J. Structure of mammalian respiratory complex I. Nature 536, 354–358 (2016).
Chang, E. et al. 18F-FAZA PET imaging response tracks the reoxygenation of tumors in mice upon treatment with the mitochondrial complex I inhibitor BAY 87-2243. Clin. Cancer Res. 21, 335–346 (2015).
Krebs, H. A. The Pasteur effect and the relations between respiration and fermentation. Essays Biochem. 8, 1–34 (1972).
Hao, W., Chang, C. P., Tsao, C. C. & Xu, J. Oligomycin-induced bioenergetic adaptation in cancer cells with heterogeneous bioenergetic organization. J. Biol. Chem. 285, 12647–12654 (2010).
Leonard, P. G. et al. SF2312 is a natural phosphonate inhibitor of enolase. Nat. Chem. Biol. 12, 1053–1058 (2016).
Muller, F. L. et al. Passenger deletions generate therapeutic vulnerabilities in cancer. Nature 488, 337–342 (2012).
Gaitonde, M. K., Murray, E. & Cunningham, V. J. Effect of 6-phosphogluconate on phosphoglucose isomerase in rat brain in vitro and in vivo. J. Neurochem. 52, 1348–1352 (1989).
Jeffery, C. J., Hardré, R. & Salmon, L. Crystal structure of rabbit phosphoglucose isomerase complexed with 5-phospho-d-arabinonate identifies the role of Glu357 in catalysis. Biochemistry 40, 1560–1566 (2001).
Sukhatme, V. P. & Chan, B. Glycolytic cancer cells lacking 6-phosphogluconate dehydrogenase metabolize glucose to induce senescence. FEBS Lett. 586, 2389–2395 (2012).
Boultwood, J. et al. Amplification of mitochondrial DNA in acute myeloid leukaemia. Br. J. Haematol. 95, 426–431 (1996).
Lagadinou, E. D. et al. BCL-2 inhibition targets oxidative phosphorylation and selectively eradicates quiescent human leukemia stem cells. Cell Stem Cell 12, 329–341 (2013).
Samudio, I. et al. Pharmacologic inhibition of fatty acid oxidation sensitizes human leukemia cells to apoptosis induction. J. Clin. Invest. 120, 142–156 (2010).
Schöckel, L. et al. Targeting mitochondrial complex I using BAY 87-2243 reduces melanoma tumor growth. Cancer Metab. 3, 11 (2015).
Zuber, J. et al. Mouse models of human AML accurately predict chemotherapy response. Genes Dev. 23, 877–889 (2009).
Wheaton, W. W. et al. Metformin inhibits mitochondrial complex I of cancer cells to reduce tumorigenesis. eLife 3, e02242 (2014).
Sancho, P. et al. MYC/PGC-1α balance determines the metabolic phenotype and plasticity of pancreatic cancer stem cells. Cell Metab. 22, 590–605 (2015).
Griss, T. et al. Metformin antagonizes cancer cell proliferation by suppressing mitochondrial-dependent biosynthesis. PLoS Biol. 13, e1002309 (2015).
Guidance for Industry on Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers (Food and Drug Administration, Rockville, MD, USA, 2005).
Kernytsky, A. et al. IDH2 mutation-induced histone and DNA hypermethylation is progressively reversed by small-molecule inhibition. Blood 125, 296–303 (2015).
Duncan, D. C. et al. Integrated genomic analyses identify ERRFI1 and TACC3 as glioblastoma-targeted genes. Oncotarget 1, 265–277 (2010).
Mueller, W. et al. Downregulation of RUNX3 and TES by hypermethylation in glioblastoma. Oncogene 26, 583–593 (2005).
Sun, Y. et al. Metabolic and transcriptional profiling reveals pyruvate dehydrogenase kinase 4 as a mediator of epithelial-mesenchymal transition and drug resistance in tumor cells. Cancer Metab. 2, 20 (2014).
Sharpley, M. S., Shannon, R. J., Draghi, F. & Hirst, J. Interactions between phospholipids and NADH:ubiquinone oxidoreductase (complex I) from bovine mitochondria. Biochemistry 45, 241–248 (2006).
Birrell, J. A., Yakovlev, G. & Hirst, J. Reactions of the flavin mononucleotide in complex I: a combined mechanism describes NADH oxidation coupled to the reduction of APAD+, ferricyanide, or molecular oxygen. Biochemistry 48, 12005–12013 (2009).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Roberts, A. & Pachter, L. Streaming fragment assignment for real-time analysis of sequencing experiments. Nat. Methods 10, 71–73 (2013).
Li, B. et al. Automated inference of molecular mechanisms of disease from amino acid substitutions. Bioinformatics 25, 2744–2750 (2009).
Yuan, M., Breitkopf, S. B., Yang, X. & Asara, J. M. A positive/negative ion-switching, targeted mass spectrometry-based metabolomics platform for bodily fluids, cells, and fresh and fixed tissue. Nat. Protoc. 7, 872–881 (2012).
Acknowledgements
We thank members of the Center for Co-Clinical Trials, Institute for Applied Cancer Science, the Glioblastoma (GBM) and AML/MDS Moon Shots for intellectual and financial support. We would especially like to express our gratitude and appreciation to C. Vellano for his help editing and assembling this manuscript; F. F. Lang and J. Gumin for providing GSC models; C. Kingsley and the MDACC Small Animals Imaging Facility; and N. Satani and E. Lin for validation of antibodies and preparation of samples for analysis, D. Bigner for D423 cells and D. N. Louis for Gli56 cells. P.Mo. was supported by The Agilent Technologies Thought Leader Award. R.A.D., A.-N.A.A., R.S., and J.Hi. were supported by The Medical Research Council (MC_U105663141 and MC_UU_00015/2 to J.Hi.). M.K., S.T., A.L., P.Ma., H.M., and Q.Z. were supported by CPRIT grant RP140218. F.L.M. was supported by the CPRIT RP140612 and National Institutes of Health (NIH) Brain SPORE P50CA127001, and A.C.S. was supported by the Research Scholar Grant RSG1514501CDD. G.F.D. was supported by the American Association for Cancer Research (AACR) 14-90-25 and by the Sheikh Ahmed Bin Zayed Al Nahyan Center for Pancreatic Cancer Grant. This work was supported in part by the Leukemia & Lymphoma Society through its Therapy Acceleration Program (TAP) and by the MD Anderson Moon Shots program.
Author information
Authors and Affiliations
Contributions
The studies were designed with input from J.R.Ma., J.R.Mo., Y.S., M.Pr., M.B., J.Ha., C.B., P.Mo., J.Hi., M.K., P.J., M.E.D.F., C.T., T.P.H., G.F.D., and F.M. In vitro experiments were performed by J.R.Ma, J.R.Mo., V.G., L.Ha., Y.T., Y.S., M.Pr., S.G., M.M., T.K., M.B., P.Mo., J.B., G.G., M.G.D., J.Ha., Y.J., T.L., H.M., P.Ma., M.Pe., R.S., T.S., M.S., V.K.H., C.C.C., and Q.Z. IACS-010759 was developed and conceived by M.E.D.F., T.M., C.C., B.C., G.L., Z.K., A.P., J.T., and P.J. In vivo studies were performed by Y-H. L., N.F., J.Ga., J.Gr., and R.M. Immunohistochemistry was performed by J. Ac., E.C., S.K., and J.R-C. Computational chemistry was performed by J.Hi. and J.B.C. Pharmacokinetic analysis was performed by S.H., Q.X., and Y.J. FACS data analysis was performed by J.R.Mo. and S.G. Glioma stem cell work was performed by Y.S., T.S., J-W.D., V.K.H, J.F.d.G., and C.C.C. Bioinformatic analysis was performed by C.B. Clinical positioning in AML was designed by M.K., N.D., J.R.Ma, M.E.D., and P.J. Metabolomic data for glycolysis deficient was generated by Y.S., C.B., and J.As. and for leukemia by J.R.Mo., S.T., A.L., and P.Mo. Stable-isotope labeling study design, analysis and interpretation was performed by P. Mo. Mouse complex I assays were designed and performed by J.Hi., A.-N.A.A., and R.S. Normal bone marrow samples were provided by S.C. and G.A-A. Writing and preparation of the manuscript and figures were performed by J.R.Ma., J.R.Mo., T.P.H., R.A.D., A.D., Y.S., M.E.D.F., M.K., P. Mo., and P.J.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–16, Supplementary Tables 1–4 and Supplementary Note
Rights and permissions
About this article
Cite this article
Molina, J.R., Sun, Y., Protopopova, M. et al. An inhibitor of oxidative phosphorylation exploits cancer vulnerability. Nat Med 24, 1036–1046 (2018). https://doi.org/10.1038/s41591-018-0052-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-018-0052-4
This article is cited by
-
Multiomics analysis identifies oxidative phosphorylation as a cancer vulnerability arising from myristoylation inhibition
Journal of Translational Medicine (2024)
-
Characterizing OXPHOS inhibitor-mediated alleviation of hypoxia using high-throughput live cell-imaging
Cancer & Metabolism (2024)
-
Transcending frontiers in prostate cancer: the role of oncometabolites on epigenetic regulation, CSCs, and tumor microenvironment to identify new therapeutic strategies
Cell Communication and Signaling (2024)
-
FGFR inhibition blocks NF-ĸB-dependent glucose metabolism and confers metabolic vulnerabilities in cholangiocarcinoma
Nature Communications (2024)
-
Chronic lymphocytic leukemia patient-derived xenografts recapitulate clonal evolution to Richter transformation
Leukemia (2024)